In the end, the electrons would all collapse into the nucleus. This radiation would send energy from the electron's movement, but it would also shrink its orbit.

According to Maxwell, accelerating charged particles release electromagnetic radiation, thus an electron rotating around the nucleus should do the same. He coined the term "orbits" to describe these circular pathways.īecause electrons are negatively charged and the nucleus is a highly concentrated mass of positively charged particles, the nucleus is held together by a strong electrostatic force of attraction.Įlectrons orbit the nucleus with predetermined trajectories, according to Rutherford. He also believed that the electrons that surround the nucleus travel at high speeds in circular paths. The nucleus is the name he gave to this part of the atom.Īccording to the Rutherford model, an atom's nucleus is surrounded by negatively charged electrons. Positively charged particles and the majority of an atom's mass were packed into a tiny space. Rutherford hypothesised the atomic structure of atoms based on the following findings.
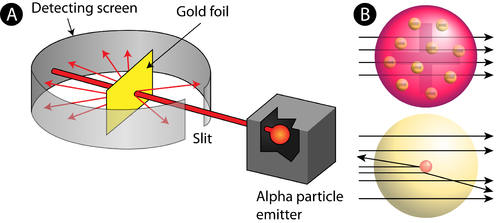
The size of the nucleus was very small compared to the total size of the atom making most of the atom largely empty. A strong electrostatic force of attraction held these two entities together bound inside the atom. The electrons revolving around the nucleus are negatively charged and the dense nucleus consisting of protons are positively charged. The atom has no net charge and is ‘electrically neutral’ as there are equal numbers of positively and negatively charged particles in an atom. The atomic nucleus is surrounded by negatively charged particles called electrons that revolved around the nucleus at very high speeds on fixed circular paths. It was later theorized and proved that the nucleus also consisted of uncharged particles called neutrons which had mass but bore no charge. This volume was called the nucleus of the atom and the rest of the atomic space was mostly empty. Rutherford’s Postulates and Their Salient Features Based on the Experimental Observationsīased on the experimental observations, Rutherford most of the mass of the atom is due to positively charged particles (later called photons) concentrated at the centre of the atom. Very few of the alpha particles were deflected back almost retracting the same path they were bombarded in, at an angle of 180 degrees which insinuated that the volume occupied by the positive charge is very minute compared to the entire atomic structure. Some of the alpha particles were deflected by the gold foil by very small angles giving rise to the conclusion that the positive charge is not uniformly distributed in the atom unlike what Thompson proposed but in reality, concentrated at a very small space most likely in the centre of the atom.

This contradicted Thompson’s model as most of the atom was observed to be virtually made of space. Observations and Conclusions from Rutherford’s Experiment He did notice that the majority of the alpha rays passed through the gold foil in a straight line and only a few reflected in different directions.Īpart from this, more observations were then obtained by Rutherford. As a source of alpha rays, he used the radioactive element polonium. Rutherford induced alpha rays to collide with a gold foil. Rutherford carried out an experiment in which he bombarded a thin sheet of gold foil with α-particles and then analysed the track of these particles after they collided with the gold foil. Ernest Rutherford, a British scientist, did an experiment and based on what he saw, he came up with a theory about the atoms of elements and came up with the Rutherford Atomic Model. Thomson’s plum pudding model didn't work well when he tried to explain some experiments about how elements have atoms. He postulated that the atom was like a Pudding with the mass and positive charge distributed across its surface and electrons embedded in the pudding-like plums. So, if there was a negative charge and the atom was electrically neutral, there had to be a positive charge. JJ Thomson proposed the Plum Pudding Model shortly after he observed the presence of negatively charged particles that he named electrons. The Atom was then considered to be indivisible, but when JJ Thompson discovered negatively charged particles in the atom, the norm was challenged, and the scientific community set out on another expedition. There have been many scientists who have proposed different models of the atom, right from the ancient Greeks and Indians. The discovery and research of quarks is a step in that direction. Originally, an atom was thought to be the smallest unit (atomic meaning unbreakable) in existence, but that theory has since been proved redundant by the discovery of subatomic particles and research is still underway to find out if these subatomic particles still can be split further. It is common knowledge today that an atom consists of electrons, protons, and neutrons.
